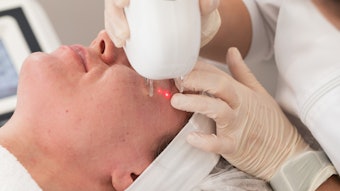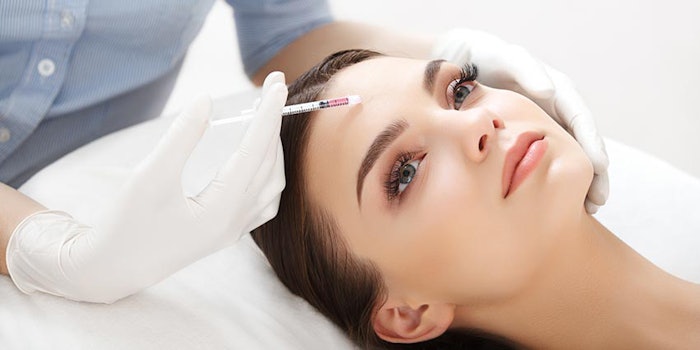
The efficiency of Botox and Xeomin, two of the most popular botulinum toxin type A fillers in the U.S., was the subject of recent studies in Dermatology Times and Cosmetic Surgery Times. The two injectables were scrutinized on their effects on glabellar, or frown lines, in three tests.
In the final, head-to-head trial, Botox and Xeomin were determined equivalent after one month, with similar results occurring in the second month after treatment, with 97% of patients satisfied at one month.
The Trials
The publications reached out to over 8,000 physicians to poll the differences found between six sets of images of patients before and 30 days after treatment, three each depicting patients injected with Botox or Xeomin. 60–65% could not distinguish which toxin had been used, while those who chanced an answer were incorrect between 24–73% of the time.
After a pivotal trial found that Xeomin was significantly effective over a placebo after 30 days, the two toxins were put head-to-head in a multicenter, randomized parallel trial including 250 women age 18–50 with glabellar lines scoring 2 or 3 on the Facial Wrinkle Scale (FWS).
Efficacy variables, clinical response by the treating physician and overall patient satisfaction were measured every month for four months; an independent masked panel of physicians determined whether results marked a ≥1 point FWS improvement from baseline at maximum frown at the primary efficacy variable.
Patients reported a median onset date at 3 days, with ≥93% reporting a peak effect within one month after treatment. 11.5% and 14.1% of patients in the Xeomin and Botox groups, respectively, experienced side effects of treatment.
Source: Dermatology Times