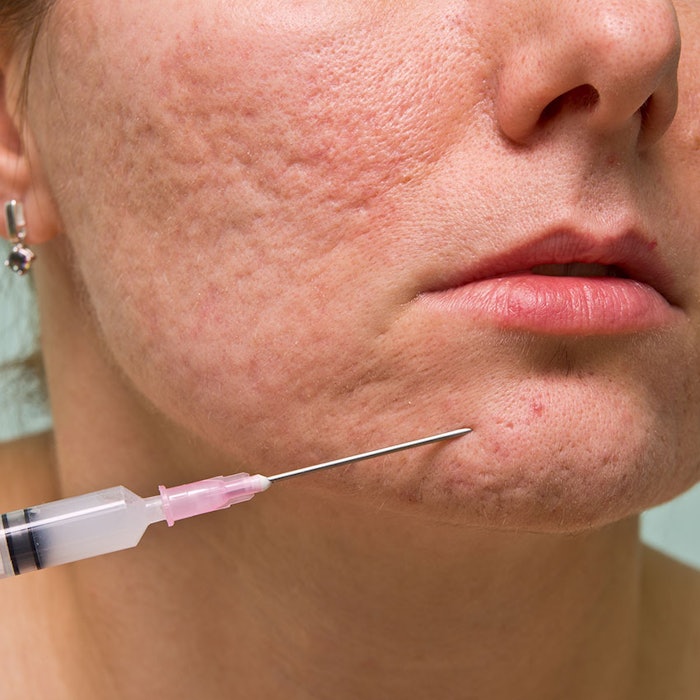
Acne is the most common skin disorder in the U.S., affecting 40–50 million people1 and up to 95% of people with acne may go on to suffer from scarring.2 In an independent study, more than 70% of respondents felt their acne scars negatively impacted their self-confidence, with 92% indicating self-confidence would be restored or improved if their acne scars were healed.3
The U.S. Food and Drug Administration (FDA) has approved the dermal filler, Bellafill® from Suneva Medical, Inc., for the treatment of acne scars. Bellafill is proven to be safe and effective for the correction of moderate to severe, atrophic, distensible facial acne scars on the cheek in patients over the age of 21 years. The treatment is an in-office procedure with little to no downtime.
References
- www.aad.org
- Layton et al. A Clinical Evaluation of Acne Scarring and Its Incidence, Clin Exp Dermatol 19:303–8 (1994)
- Formula PR Independent Study, 2013










