An approved skin cancer treatment for patients with advanced melanoma, pembrolizumab has now verified its clinical benefit with two new trials.
On December 18, 2015, the U.S. Food and Drug Administration (FDA) expanded the label to include the approval of pembrolizumab (Keytruda Injection, Merck Sharp & Dohme Corp.) for the treatment of patients with unresectable or metastatic melanoma. This expansion now includes the initial treatment of patients with unresectable or metastatic melanoma with pembrolizumab.
The recommended dose and schedule for pembrolizumab is 2 mg/kg Q3W administered as an intravenous infusion every three weeks until disease progression or unacceptable toxicity. This application was granted priority review. The development program for pembrolizumab for this indication received orphan drug designation and was granted breakthrough therapy designation.
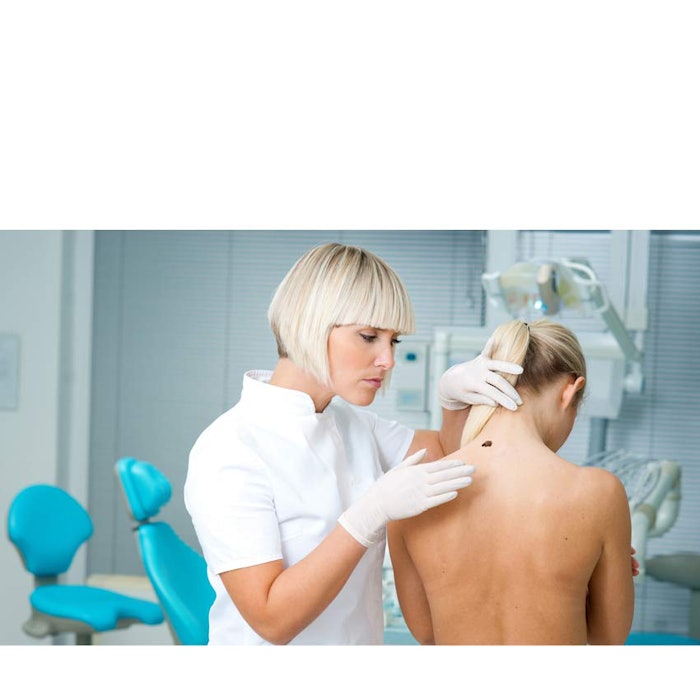
Within the last five years, targeted therapy and immunotherapy have emerged as viable treatment options for patients with advanced melanoma. Although these therapies have promising implications, early detection still gives patients the best chance of survival.
Read more about skin cancer treatments and early detection in Skin Inc.