Cryosurgical product manufacturer CryoConcepts, LP has received FDA clearance on two patent-pending crysurgical devices: CryoTouch and CryoLab.
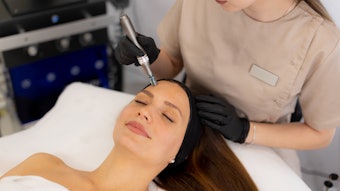
A portable cryosurgical device that delivers cryogen to lesion sites, CryoTouch can use either applicators or cones, based on the physician’s preference; CryoLab is a benchtop unit that delivers cryogen for medical practices that perform a high volume of cryosurgery.
The platforms have been cleared to treat up to 23 different indications.
“We are excited to get these clearances from the FDA,” said Sam Niedbala, Ph.D., CEO of CryoConcepts. “CryoTouch and CryoLab have been designed to address user needs in a product category that has not seen innovation in over two decades. Our new, patent pending, products will deliver greater efficacy and are much easier-to-use. These new products position CryoConcepts as the only company that is a one-stop source for cryosurgical products ranging from disposable devices to capital equipment.”
The majority of the cryosurgical market consists of two types of products—liquid nitrogen-based delivery systems, primarily used by dermatologists, which require frequent refills due to evaporation; and portable, disposable can-like devices used by pediatricians and general practitioners. “Our new products are easy-to-use and deliver more effective treatments while being highly efficient,” said CryoConcepts CMO Bill Hinchey.
The company, which currently markets cryo devices under the CryOmega, CryoClear and CooLifting brand names, plans on a series of “aggressive introductions into a variety of professional medical markets” over the course of 2019.