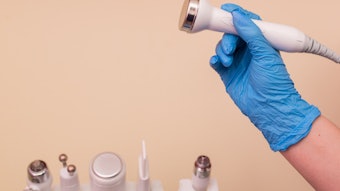
Health Canada has not approved any plasma pen, or "fibroblast" devices, for sale in Canada based on their belief that it may pose health risks. Plasma pens are used for cosmetic skin treatments like eyelid lifts, wrinkle and scar reduction and mole and skin tag removal. They focus electricity on the surface of the skin, which causes a controlled burn and spreads heat through the targeted area.
These pens can be used in spas as well as sold to consumers online and through esthetician training courses, which Health Canada is aware of. In June 2018, Health Canada contacted a number of manufacturers, distributors and importer to inform them that selling or advertising these devices was illegal in Canada without the appropriate licensing.
Health Canada has also moved to start contacting spa professionals to warn them of the side effects and negative impacts of this device and inform them about the medical device licensing requirements associated with plasma pens. The side effects of these pens, even when used properly, include pain, swelling, redness, sagging skin, hyperpigmentation, UV sensitivity and skin peeling or crusting. These side effects increase with the duration, frequency and intensity of the treatments, as well.
Check whether medical devices have been authorized for sale by searching Health Canada's Medical Devices Active License Listing or Recalls and Safety Alerts Database.