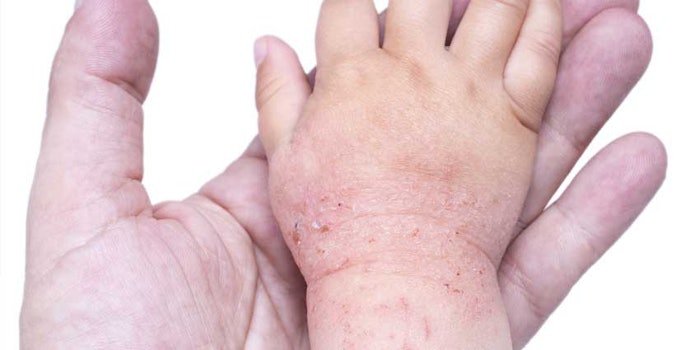
The U.S. Food & Drug Administration (FDA) has approved Eucrisa (Anacor Pharmaceuticals, crisaborole), a treatment to benefit those suffering from eczema.
Applied twice daily, the treatment is a phosphodiesterase 4 (PDE-4) inhibitor, helping with mild to moderate eczema in patients 2 years old or order. According to the FDA, the method of how Eucrisa treats atopic dermatitis is not known.
Elaine C. Siegfried, M.D., professor of pediatrics and dermatology at Saint Louis University, Cardinal Glennon Children's Hospital, St. Louis, Missouri explained how the corticosteroid-sparing topical option is welcomed.
“The other two alternatives (pimecrolimus cream and tacrolimus ointment) carry black box warnings and labeled limitation on duration of use,” she explained. “Although most pediatric dermatologists prescribe these medications in infants and children without long-term safety concerns, prescribing Eucrisa is not hampered by this medicolegal burden. However, cost and access could be a limitation.”
FDA Approvals and Safety
While this is the first approval for a common disease, which does not seem to get enough attention, Adam Friedman, M.D., associate professor of dermatology and director of translational research in dermatology at George Washington School of Medicine and Health Sciences is hopeful for other therapies to be approved.
“I envision crisaborole being used in a similar manner to calcineurin inhibitors, both as proactive treatment for affected delicate areas like the eyelids, face, body folds, groin or mild disease elsewhere,” he said. “But, more importantly, [I envision it] as preventative maintenance therapy for disease areas that recur frequently after topical steroid use has been discontinued (though without the baggage of a black box warning and possible substance P-induced burning at the onset of use).”
Despite the new approval, Friedman encourages patients and professionals who work with clients who have eczema should continue to be educated. There are multiple ways to properly manage the chronic condition such as proper soap, moisturizer and treatment use and proper education on treatment safety myths.
The Foundation of the FDA’s Approval
Two placebo-controlled trials studying 1,522 people from ages 2-79 with mild to moderate atopic dermatitis were the root to the FDA’s approval for Eurcrisa. After 28 days of treatment with Eurcrisa, those in the active group had clear or nearly clear skin.
While the treatment’s outcome is successful, some negative reactions such as hypersensitivity are possible and those who have this reaction to crisaborole should not use Eurcrisa. Applying to those with this reaction can cause pain, burning or stinging.
Source: Dermatology Times










