Editor’s note: This article originally was published in the September 2006 issue of Cosmetics & Toiletries magazine, and has been reprinted with permission.
The approach to ethnic skin care and treatments can be challenging. This mainly is because ethnic skin has a propensity to develop hyperpigmentation—or a darker discoloration of the skin—or hypopigmentation—a lighter discoloration of the skin—when the appropriate products or treatments are not utilized. To further examine the challenges of ethnic skin care, one must first define ethnic skin.
Defining ethnic skin
The Fitzpatrick Skin Type classification often is used to define ethnic skin. This scale ranges from type I (ivory white skin) to type VI (dark brown skin) and identifies skin type based on its reaction to UV light. Skin of color can be classified as skin types IV–VI.
Biology of ethnic skin
Pigmentation in the skin is determined at the cellular level. Melanocytes are the cells that make melanin, or pigment, in the skin. Melanosomes are the packages of melanin produced by the melanocytes. Pointy extensions of the melanocytes then transfer melanosomes into the keratinocytes.
Although there may be some variation in the number of melanocytes among races, this difference is not striking. In all races, there are approximately 2,000 epidermal melanocytes/mm2 on the head and forearms, and 1,000 epidermal melanocytes/mm2 on the rest of the body. These differences are present at birth.1 Thus, all people have the same total number of melanocytes; it is the distribution of melanosomes in the keratinocytes that correlates with skin color. In white skin—typically types I–III—melanosomes are small and aggregated in complexes. In black skin—types V and VI—there are larger melanosomes that are distributed singly within keratinocytes.2
The role of melanin in the skin is to absorb and scatter energy from UV light to protect the epidermal cells from damage. Melanin provides considerable protection from sun damage, and the degree of protection corresponds directly to the degree of pigmentation. This sun protection offers significant prevention of photo-aging, which is one of the primary cosmetic concerns in Fitzpatrick skin types I–III.
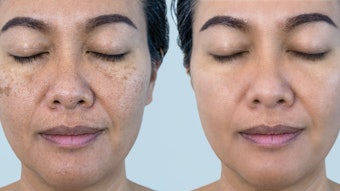
The cosmetic advantage of skin types IV–VI is an increased protection from photo-aging. However, the cosmetic disadvantage of skin of color is its propensity to develop hyperpigmentation or hypopigmentation, which ethnic clients frequently experience and are most concerned about preventing.3 Essentially, any inflammation or injury to the skin can be followed immediately by an alteration in pigmentation.
Hyperpigmentation in ethnic skin
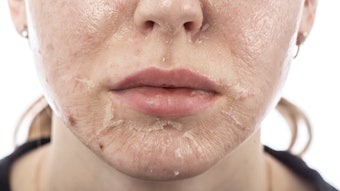
Any ingredient that has a potential to cause significant irritation or dryness when applied to the skin can cause pigment alteration in ethnic skin. Although hypopigmentation can occur, hyperpigmentation is most common. The patients at greatest risk are those with dry, sensitive skin. The most common topical treatments associated with pigment alterations include benzoyl peroxide, retinoids, salicylic acid and glycolic acid. Additionally, in predisposed clients, hydroquinone can be irritating and can lead to hyperpigmentation. Typically, individuals first report dryness and irritation. In more severe cases, this is followed by redness and even superficial erosions.
In skin types I–III, if any reaction occurs, the skin typically will return to normal once use of the product is discontinued. Ethnic skin differs from the lighter skin types because, in these clients, dryness and irritation frequently are followed by postinflammatory hyperpigmentation. In predisposed individuals, hyperpigmentation usually develops within one to two weeks and can last for several months. However, it is important to note that pigment alterations are rare when benzoyl peroxide, retinoids, salicylic acid and glycolic acid are utilized correctly in ethnic skin. Clients with ethnic, dry, sensitive skin can avoid pigment alterations by using products that are formulated in emollient-based vehicles or with lower concentrations of the active ingredient.
Retinoids
Acne is the most common dermatologic diagnosis, with an incidence of up to 29% among black patients.4 In comparative studies between black and white patients, the prevalence of acne appears to be equal.5
The first line of therapy for acne continues to be retinoid therapy. Adapalene, tretinoin and tazarotene are the most commonly used prescription retinoids for treating the condition. Retinol is available over-the-counter (OTC) and is used primarily in anti-aging formulations.
Adapalene has been well-studied in the treatment of acne for black clients. It is best-suited for those with mild to moderate acne. Of the retinoids, it is useful especially in those with dry, sensitive skin because of its low incidence of irritation.6 Studies of adapalene in black clients with acne also report reductions in hyperpigmented macules—or patches of skin altered in color—and density of hyperpigmentation.7
Tretinoin also has been studied as a treatment for acne in black individuals. It is best-suited for those with moderate acne and oily skin. Similar to adapalene, tretinoin is effective not only on comedones, but also on the associated hyperpigmentation.8
Tazarotene is an excellent treatment choice for clients with moderate to severe acne. Individuals must be advised to expect dryness and scaling when beginning this medication. Skin care professionals often recommend a dosage of tazarotene be applied every other night for the first two weeks, then be increased to a nightly application in order to improve tolerability. As with all topical retinoids, among some skin types, irritation can lead to hyperpigmentation.
Benzoyl peroxide
Benzoyl peroxide is available in both OTC and prescription formulations. It is quite effective in treating acne; however, there are a few side effects to be aware of in skin of color. Skin of color can vary in pigment from light brown to dark brown, but it is generally falls among types V–VI on the Fitzpatrick scale. Some formulations of benzoyl peroxide can leave a white or gray film, making it cosmetically less acceptable for patients with skin of color. Additionally, when significant irritation occurs, some clients can develop hyperpigmentation with benzoyl peroxide. Overall, however, benzoyl peroxide is well-tolerated and is a useful adjunct in treating acne in ethnic skin.
Salicylic acid
Salicylic acid is common in many OTC acne formulations in concentrations of 0.5–2.0%. Irritation and postinflammatory hyperpigmentation in predisposed consumers usually are observed with higher concentrations. Salicylic acid also can be used as a chemical peel for patients with acne and hyperpigmentation. It is well-tolerated in concentrations of 20–30% in consumers with oily acne-prone skin. Salicylic acid peels do not need to be timed or neutralized, and are applied every two to four weeks for a series of five treatments.
Glycolic acid
Glycolic acid also is common in many OTC acne and anti-aging formulations. Formulations of up to 10% can be found in anti-aging creams and acne treatments, and up to 20% in some cleansers. It is important to note that the efficacy of glycolic acid is based on the proportion of free acid in a given product. The amount of free acid can be determined from the pH level and the concentration of glycolic acid in a product. Thus, when comparing two products with the same percentage of glycolic acid, the pH is very important. A product with a very low pH will be more irritating but will have a greater amount of free acid and, thus, will be more efficacious at increasing cell turnover and exfoliation. A product with a higher pH will be gentler and have humectant properties but will not be as efficacious in cell renewal.9
Glycolic acid also is utilized as a superficial chemical peel. Glycolic acid peels at 20–30% can be used safely on ethnic skin, but they need to be timed and neutralized 2–3 minutes after application. Higher-concentration peels have a greater risk of causing hyperpigmentation in ethnic skin—types IV–VI—because these peels can be significantly deeper. As with salicylic acid, repeated treatments are required in order to achieve the best results.
Hydroquinone
Hydroquinone is the treatment of choice for hyperpigmentation in the United States. Formulations at 2% are available OTC; 4% formulations are commercially available with a prescription. Often, compounded formulations of 6–10% are required in clients with ethnic skin. Most people with ethnic skin tolerate hydroquinone well. However, there is a small percentage of people with hydroquinone sensitivity who develop irritation and hyperpigmentation with hydroquinone use. In these clients, the degree of irritation increases with the ingredient concentration.
Another common issue with hydroquinone is its tendency to oxidize and turn brown when exposed to the air. Hydroquinone packaged in jars frequently turns brown within weeks, once opened. It must be stabilized and packaged in tubes or pumps in order to avoid this problem.
Although hydroquinone has been used for more than 40 years in the United States as a skin-lightening agent, OTC formulations of the ingredient are banned in Europe and Asia due to reports that it was carcinogenic in animal studies.
Ochronosis is a rare idiopathic darkening of the skin that has been reported with prolonged hydroquinone use. Most cases occurred in South Africa where hydroquinone and other skin-bleaching agents were used daily to lighten facial skin. Hydroquinone has been banned in South Africa due to the high incidence of ochronosis in that country. Interestingly, reports of ochronosis are rare in the United States. Ochronosis most often is associated with prolonged daily application of hydroquinone to the entire face for a period of 20–30 years. For this reason, most dermatologists recommend that the ingredient be applied only to the affected area and that its use be discontinued once improvement has been achieved.
Skin-bleaching agents
Many other skin-bleaching agents can be used to lighten the skin. These include mequinol, azelaic acid, kojic acid, arbutin, soy, vitamin C, licorice extract and niacinamide. Mequinol is used widely in Europe as a skin lightener. It is the monomethyl ether of hydroquinone, and was introduced recently in the United States as a prescription skin-lightening agent. Azelaic acid 15–20% requires a prescription and has been found comparable to 2% hydroquinone in studies.10 Kojic acid is available OTC in formulations of 4% and also is comparable to 2% hydroquinone.11 Soy and arbutin are available in many OTC products as skin-lightening agents. Although the list of skin-bleaching agents is extensive, hydroquinone remains the gold standard. Significant research in this area is still is needed in order to find equally effective alternatives.
Additional considerations
Moisturizers are an extremely important component of ethnic skin care. Patients with darker skin types often develop a gray appearance to their skin when it is dry. This phenomenon frequently is referred to as “ashy” skin. In fact, some studies suggest that transepidermal water loss is greater in black patients.12 This finding may account for the high incidence of dry skin seen clinically in black clients. Highly effective moisturizers often are required to treat this ashy appearance. In more severe cases, moisturizers with ingredients such as lactic acid or salicylic acid are most effective.
Another common concern for clients with ethnic skin is the gray tint observed with the application of topical sunscreens that contain zinc oxide or titanium dioxide. Similarly, foundations and makeup that contain physical blockers tend to cause this gray/blue tint or hue to the skin. This gray sheen is most prominent in types V and VI skin, and can make certain sunscreens cosmetically unacceptable for ethnic skin.
Micronized formulations have made these physical blockers more cosmetically acceptable. Other solutions include combining chemical blockers with physical ones so that a lower concentration of the physical blocker is required in order to achieve the desired SPF. Despite these advances, developing a purely physical blocker with a high percentage of zinc oxide or titanium dioxide that does not leave a gray hue or tint on darker skin types remains a challenge.
Additionally, other topical products that leave any type of residue are not cosmetically acceptable in ethnic skin. This is because a thin white residue that is transparent in lighter skin can be extremely obvious in darker skin tones. This phenomenon has been observed in some urea and benzoyl peroxide products. However, it can occur in any topical product that leaves a powdery residue when it dries on the skin.
Summary
The cosmetic concerns of ethnic skin are unique and varied. Skin sensitivity appears to be similar among different racial ethnic groups.13 However, the aftereffects results of skin irritation in darker-skinned persons are more pronounced. Although most products are well-tolerated in ethnic skin, it is important to understand the unique features of this ethnic skin type when recommending treatments.
References
1K Jimbow, WC Quevedo, G Prota, TB Fitzpatrick, Biology of melanocytes, in Dermatology in General Medicine 5th ed, IM Freedberg, AZ Eisen, K Wolff, KF Austen, LA Goldsmith, SI Katz, TB Fitzpatrick et al., eds, New York, NY: McGraw Hill 192–220 (1999)
2IK Rosdahl and G Szabo, Thymidine labeling of epidermal melanocytes in UV-irradiated skin, Acta Dermato-Venereologica 56(2) 159–61 (1976)
3PE Grimes, Skin and hair cosmetic issues in women of color, Dermatologic Clinics 18(4) 659–65 (2000)
4P Dunwell and A Rose, Study of skin disease spectrum occurring in an Afro-Caribbean population, International Journal of Dermatology 42 287–9 (2003)
5RM Halder, PE Grimes, CL McLaurin, MA Kress and JA Kenney Jr., Incidence of common dermatoses in a predominantly black dermatologic practice, Cutis 32 388–90. (1983)
6J Czernielewski, M Poncet and F Mizzi, Efficacy and cutaneous safety of adapalene in black patients versus white patients with acne vulgaris, Cutis 70(4) 243–8 (2002)
7WK Jacyk, Adapalene in the treatment of African patients, Journal of the European Academy of Dermatology Venerol 15 supplement 3 37–42 (2001)
8SM Bulengo-Ransby, CEM Griffiths, CK Kimbrough-Green, LJ Finkel, TA Hamilton and CH Ellis et al., Topical tretinoin (retinoic acid) therapy for hyperpigmented lesions caused by inflammation of the skin in black patients, The New England Journal of Medicine 328 1438–43 (1993)
9DT Sepp, The ABC’s of Alpha-Hydroxy Acids, Les Nouvelles Esthetiques, 13(12) 30–36 (1998)
10VM Verallo-Rowell, V Verallo and K Graupe, et al., Double blind comparison of azelaic acid and hydroquinone in the treatment of melasma, Acta Dermato-Venereologica Supplementum 143:58–61 (1989)
11A Garcia and JE Fulton Jr., The combination of glycolic acid and hydroquinone or kojic acid for the treatment of melasma, DermatologicSurgery 22(5) 443–7 (1996)
12N Wesley and HI Maibach, Racial (Ethnic) Differences in Skin Properties: The objective data, American Journal of Clinical Dermatology 4(12) 843–860 (2003)
13AV Rawling, Ethnic skin types: are there differences in structure and function, International Journal of Cosmetic Science 28 79–93 (2006)