Vitamin A, or retinol, is one of the most effective and versatile treatment modalities available to the skin care professional. It can be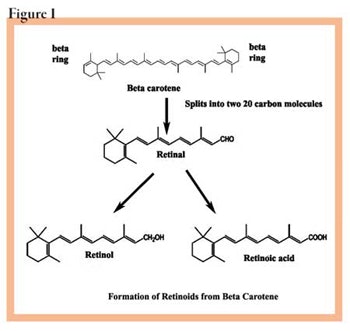
The chemistry
Vitamin A is most commonly found in nature as an alcohol form, but only in animals. Carotenoids that contain vitamin A, such as beta-carotene, are found in plants such as dark or yellow vegetables, and carrots. These are precursors of vitamin A. If anyone tries to sell you a “natural plant vitamin A,” there is no such chemical. In some plants, such as carrots, carotenoids make up a large group of compounds. As you can see in Figure 1, beta-carotene is the precursor in the plant of vitamin A.
There are four major forms of carotenes based on the ring 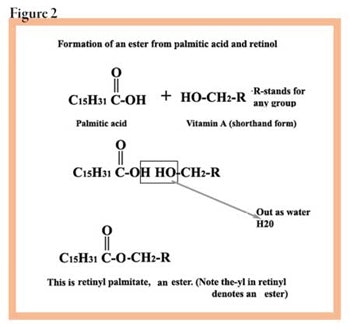
More importantly, notice in Figure 1 how vitamin A is produced from beta-carotene by animals. The point is that vitamin A comes in many forms, all of which must be made chemically, either in animals or manufactured otherwise. For example, retinol is converted to retinyl palmitate and stored in the liver. You can buy retinyl palmitate from several manufacturers as an ingredient, or you can buy it as
a finished skin care product.
Chemical forms of vitamin A
The common, readily available forms of vitamin A include retinol, retinyl palmitate, retinyl acetate and retinoic acid. Retinol and the other two compounds known as esters—the palmitate and the acetate—will be discussed.
A fourth form—the aldehyde form, retinal—is available and will be touched on briefly since it is new and not fully evaluated at this time.
Retinol. Retinol is the most common form of vitamin A. It is the 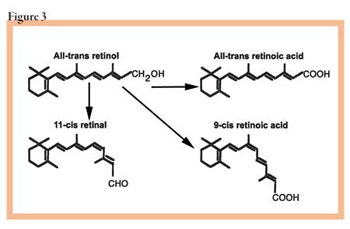
Retinyl palmitate. This is an ester, which is a special chemical formed when an alcohol is combined with an organic acid. Most perfumes are based on the use of esters. Retinyl palmitate is formed when retinol, an alcohol, is combined with palmitic acid, a 16-carbon fatty acid. When combined and heated, the alcohol group on the vitamin A combines with the acid group on the fatty acid. This is diagrammed in Figure 2.
Retinyl acetate. The two most common esters of retinol are palmitic and acetic. Retinyl acetate is formed by combining retinol with acetic acid.
Retinoic acid. The acid form of vitamin A is called retinoic acid, and is available by prescription only. It is the form of vitamin A used by the body at the cellular level. See Figure 3 to see the forms of vitamin A.
Vitamin A and the body
Treatment ingredients are used because the normal body level is low and you are replacing what is missing, or you are adding a substance to modify or change an existing condition that is not desirable. If the body had everything it needed and every system was running perfectly normal, each person would be great, beautiful and perfectly healthy. Sadly, this is not the case; people need help, and one of the substances that can help is vitamin A.
To understand what vitamin A actually does in the skin in order to be able to use it advantageously, examine the pathway from food intake to the cellular use of vitamin A.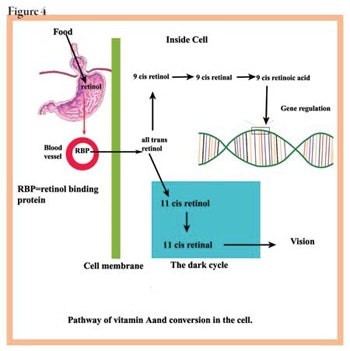
Vitamin A intake. Most of the vitamin A in the body is from food, and is mainly from beta-carotenes, which are converted to vitamin A in the intestine and liver, although a portion is absorbed as beta-carotene from the intestines into the lymphatics, passing from there into the circulatory system. The beta-carotene is split into two molecules of retinol, which is then enzymatically changed into two other compounds.
First, retinol is oxidized to form retinal, which is used to make rhodopsin for the eyes. Then retinal is oxidized to generate retinoic, which binds to certain proteins in the cytoplasm of the cells—a step necessary to allow it to enter the nucleus and bind with DNA. It is the action of retinoic acid on the DNA that accounts for all the biological effects on the skin. Figure 4 outlines this pathway in a simple form.
Isomerization of the retinoids produces two major chemical formsb. One type, known as 11-cis-retinal, is the chromophore that is part of the visual pigment in the eye. The other one makes up the hormonal retinoids, which are known as all-trans and 9-cis retinoic acid. It is this form of retinoic acid that regulates the expression of cellular genes via the activation of the two classes of nuclear retinoid receptorsc. These receptors are known as retinoic acid receptors (RARs) and the other is called the retinoid X receptors (RXRs). Ultimately, these forms of retinoids carry out many physiological processes, such as reproduction, embryonic development, postnatal growth, differentiation and maintenance of various epithelia, certain functions of the immune response, and, of course, vision. At this time, little is known about what regulates the uptake of retinoids by cells.
Vitamin A and the skin
Do retinoids actually get into the skin, and, if so, how much? The answer: yes, they get into the skin—although not much—and they enter through several routes. This is yet another mystery because the mechanism of transcutaneous absorption of topical retinoids is not well understood, just as the cellular uptake is not fully explained. Retinoic acid will be the model examined because it is the most studied of the retinoids.
When applied to the skin, all-trans retinoic acid first undergoes isomerization, but this happens to only part of the applied amount. It goes from 9-cis to 13-cis. See Figure 3. Only about 20% will penetrate the skin, with 80% remaining on the surface. How much additional retinoid goes into the skin depends on a number of factors; mainly, how the product is formulated and the amount of hairy surface to which it is applied.1 Less than 5% of a cream-based retinoic acid will penetrate the skin in 30 minutes. This is odd because when retinoids are tested in an in vitro model on excised skin, penetration is fairly rapid.
It is known that after a retinoid—such as retinol—is in the skin, it binds first to retinol-binding protein, then to specific cellular-binding proteins within the cell, known as CRBP-I and -II. Retinol binds to CRABP-I and retinoic acid binds to CRABP-II, respectively. These proteins act as both storage and as shuttle proteins within the cytoplasm, and, at the same time, serve to regulate the concentration of the retinoids within the cell. They both bind and release retinoids according the needs of the cells. Keep in mind that the ultimate goal of retinoids is to react with the nuclear DNA and program it for a specific action.2 One note: 13-cis retinoic acid is not bound by CRABP-I/-II or any other binding proteins in the cytoplasm. As a result, it is free to enter the nucleus once it is in the cell. This may explain why this form of retinoic acid is so irritating and teratogenic if taken orally or even applied topicallyd.3
Retinol and retinol esters. Ask yourself, “Why am I using vitamin A? Does the skin need this vitamin—does it have enough or too much vitamin A. What am I trying to do with this ingredient?” These are fundamental questions that all too few skin care professionals ever raise, even if they are conscientious about treating their clients. Understand that vitamin A is an essential vitamin—people do not and cannot make it—yet the body needs it. The amount the body requires is tightly regulated, so when it is applied, how much is getting into the skin?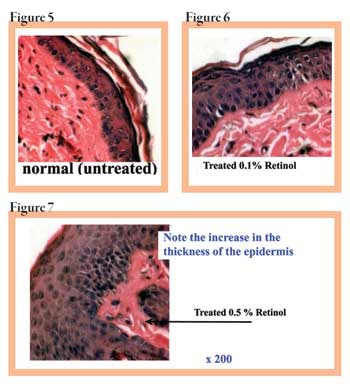
Penetration is dependant on many things, including how the product is formulated. You buy vitamin preparations by the percent of vitamin they contain, but the body uses it in terms of international units, or IU, which are measured in terms of biological activity. One IU of vitamin A is equal to 0.3 micrograms of vitamin A. So, 1 gram of vitamin A would equal 1 million micrograms/0.3 = 3.33 million units of vitamin A. Just for fun, let’s see how many IU are in a 1% vitamin A preparation. A 1% solution contains 1 gram of a substance, consisting of 1 million micrograms. Our 1% vitamin A product, assuming it is retinol, contains 3.3 million units of vitamin A. This is an enormous amount. The major biological activities of retinol are the control of cellular morphogenesis, epithelial cell proliferation and differentiation, immune modulation, stimulation of the creation of new blood vessels, and production of collagen. Some other functions include inhibition of carcinogenesis; absorption of UV light; antioxidant functions; pigment control; and, because retinoids contain several conjugated double bonds, they can trap free radicals; and while absorbing UV radiations from the sun, they protect DNA, lipid membranes and proteins from UV-induced oxidative stress. However, vitamin A in all forms is destroyed by UV light—both in the UVB (290–320 nm) spectra and UVA (320–400) spectra.
Applying various concentrations of retinol on skin produces a dose-response effect; that is, the higher the dose applied to the skin, the greater the effect on the skin. In Figures 5, 6 and 7, you can see the effects. Figure 5 is the control, or normal, untreated skin. Figure 6 is the effect of 0.1% retinol applied daily for seven days. Notice that the epidermis is thicker and the stratum corneum is less prominent. In Figure 7, you see the effect of 0.5% retinol, again applied daily for five days. There is a great deal of cellular activity with a marked increased in the size of the epidermis. This is called epidermal hyperplasia. Notice that the stratum corneum is also completely gone, due to exfoliation. Sometimes the stratum corneum will be compacted and appear thin. These histological examples illustrate the need to know not only the effect of retinol, but also to appreciate the importance of being familiar with the dosage you are applying.
There is a feedback relationship between the epidermis and the dermis. As the epidermis grows, the dermis must increase the support elements of collagen and elastin—most likely, this is direct action on the fibroblast by retinol and/or retinoic acid. Fibroblasts seem to control the rate of proliferation in the epidermis. The final answer remains to be discovered, but what is known is that certain retinoids will impede collagen synthesis, while others will stimulate collagen formation. It remains a very complex question.
Even less is understood about the effect of retinoids on the production of melanin pigment. It is known that hydrocortisone steroids plus retinoic acid and hydroquinone will reduce pigmented spots after 3–6 months of treatment. Retinoids inhibit the growth, but enhance the differentiation of melanoma cells in tissue culture. Yet, they inhibit pharmacologically, as well as hormonally induced melanogenesis in these cells. This data was developed 14 years ago, but no definitive data is available to explain the effect of retinoic acid on normal human melanocytes. It appears that retinoic acid has little effect on normal melanogenesis when studied in normal human melanocytes in tissue culture in vitro. One study suggested that the changes in the shape of the melanocytes may indicate that retinoic acid targets the cytoskeleton proteins.4
Clinical applications of retinoids
Vitamin A can be used to treat several clinical conditions, including acne, aging skin and hyperpigmentation. Each of the forms of vitamin A—retinol, retinyl acetate and retinyl palmitate—has specific reasons why it would be selected for a particular treatment.
Acne vulgaris. This is a disorder of the sebaceous follicles that is the scourge of teenagers and young adults, both female and male. Most skin care professionals understand that the major pathogenic include hyperkeratinization, which blocks sebum excretion and normal exfoliation due to infundibular obstruction; androgenic stimulation of sebaceous gland secretion; Propionibacterium acnes in the hair follicle, which produce fatty acids and cause inflammation; and a genetic component. Although the full story about how acne is transmitted is not known, a family history of acne is associated with an early onset of acne, more severe acne and often treatment-resistant acne.5
One of the problems associated with hyperkeratosis is the increase in desmosomes and tonofilaments seen in keratinocytes in comedones. Recently, substance P, a neuropeptide, was found to stimulate sebaceous gland cells, increasing the size of these cells. As you may know, substance P can be elicited with stress and, perhaps this could explain “the giant date pimple.” The social problems of the acne patient only serve to add to the severity of the condition.
Treatment. Although no one has demonstrated the conversion rate of vitamin A to retinoic acid, I believe it is about one-tenth the strength of retinoic acid, based on my observations of comparative studies of retinol and retinoic acid on mouse skin using the degree of proliferation of the epidermis as an endpoint. The final goal of any acne treatment program should be to heal the acne and restore normal skin, prevent scarring and attempt to reduce the psychological stress associated with acne. Obviously, a good treatment program that clears the acne lesion can reduce social stress a great deal.
Skin care professionals should feel comfortable and competent when treating acne that consists of comedones and pustules without cysts. A cyst, which can produce scars, is most often a sign of trouble and should be referred to a physician. See Guidelines for Treating Acne for recommendations on how to treat the issue.
I prefer retinyl acetate for the vitamin A form in acne as it penetrates faster and is less irritating than retinol; however, they all work well. Recently, retinaldehyde has become available and will probably be the retinoid of choice for treating acne since it has all the other actions of retinol, but it is an effective germicide, as well. This is most likely due to the carbonyl group (the aldehyde group) at the end of the molecule.
Aging skin. Unfortunately, there are many treatments for aging. Without question, the greatest boon to aging treatment methods was the introduction of retinoic acid by Albert M. Kligman, MD, PhD, in 1986. Since then, the world of vitamin A has exploded and many, many new compounds have been developed to treat a host of skin diseases. Aging skin still ranks as the No. 1 problem for most women.
No one really knows the whole story about aging. It is known that some genetic components are at work, as well as many environmental factors, some internal factors, such as wear and tear, and a lot of metabolic and dietary factors. It is interesting that individuals are not programmed to die at a specific set point, even though maximum life span is now thought to be 122–125 years. Quite a bit is being learned, however, because people are living longer and are healthier than ever. What is considered middle age has jumped 15–20 years during the past 50 years, and will continue to rise. One reason that vitamin A is so important as a factor in aging is the critical role it plays in skin health, but vitamin A is rapidly depleted by UV light—both UVA and UVB—so most who are exposed to UV light even indoors, such as fluorescent lighting, will have quite a lot of the vitamin A in their skin destroyed daily. For some reason, this factor in aging is not mentioned very often by aging experts.
When examining all these factors, no one ingredient is going to solve all the aging skin problems. A composite approach is needed and should include several ingredients in a comprehensive treatment program. Vitamin A is needed, but it requires other active ingredients, as well.
Treatment. There is no need to ever to get a wrinkle and wrinkles are mainly sun induce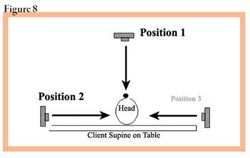
Photograph the client sitting up—get a good high-resolution camera that will allow for great definition close up. Photograph front and both side views. Next, have client lie flat for 20 minutes. Photograph the client with the camera perpendicular to her face; that is, immediately above her face, and then photograph both sides again with the camera at a right angle to her face. See Figure 8. Compare the lines in the face, around the eye, the cheek areas, the nasolabial fold and the labiomental fold. If the wrinkles are effaced by 50% or more, you can get a good result. If there is no change, the wrinkles are deep and refractory to treatment, and it will take no less than a very deep peel plus a face lift to improve this condition. See Guidelines for Treating Aging Skin.
Keep in mind that the amount of product placed on the skin will not all penetrate into the skin. In fact, only a small amount will penetrate and that will need to be converted to retinoic acid, which is further reduced. Although there is no hard and fast rule and little data to back it up, I use the rule of one-tenth. That is, about one-tenth of the amount of vitamin A, as retinol or retinyl acetate placed on the skin will become retinoic acid. It may be even less than this, but my studies have shown that a 1% vitamin A will give about the same hyperproliferative effect on the epidermis as 0.1% retinoic acid. It is possible to use 1% and 2% vitamin A product on the skin after you have had training in the use of lower doses, but do not use these higher levels without additional training. Remember that vitamin A is a hormone and, at high levels, can be systemically toxic, even when used topically.7
Hyperpigmentation. Hyperpigmentation disorders of the skin are quite common. The most frequent forms include melasma, lentigines and postinflammatory hyperpigmentation. Pigmention problems are often difficult and require a lengthy treatment program. Melasma, for example, is not fully understood—although it is associated with hormones and UV exposure—but there appear to be more factors associated with this condition.
When retinol is used in high concentrations, that is above 0.5%, irritation will occur, manifested by flaking and erythema. Retinol does not seem to be effective at low concentrations for pigmented lesion. See Guidelines for Treating Hyperpigmentation.
Getting familiar with retinoids
Vitamin A is one of the best therapeutic agents available to a skin care professional. Knowing the chemistry and the physiology of retinoids is extremely helpful when applying them for the treatment of acne, aging skin and hyperpigmentation. Vitamin A comes in several forms lumped under the term retinoids. This includes retinol, the natural alcohol form; retinal, the aldehyde form used in the biochemistry of vision; and retinoic acid, the acid form that binds to the nuclear receptor and programs the biological effects via DNA. The alcohol can be units with fatty acid to make both an acetate ester, known as retinyl acetate and palmitate ester—retinyl palmitate. The most irritating form is retinoic acid and the least irritating form is retinyl palmitate.
All the retinoids have essentially the same effect, which includes cellular differentiation, proliferation, morphogenesis and immune stimulation. In addition, retinal is germicidal, as well as being essential for vision. In the treatment of aging skin, retinyl acetate is very effective and less irritating than retinoic acid. The retinoids are both powerful and effective in treating acne and hyperpigmentation. The skin care professional needs to be familiar with the chemistry and biological actions to use retinoids both effectively and safely.
FOOTNOTES
a. The term retinoids is a general chemical classification that refers to all forms of vitamin A: retinol; retinal; retinoic acid; all the esters, such as retinyl palmitate; and all the synthetic prescription forms.
b. Isomerization is a chemical process in which a molecule changes its shape, but has the same number of atoms. In other words, the molecule weight and number of atoms is exactly the same.
c. The designation -cis and -trans in the formula denotes the presence of chemical entities on the long chain that are on the same side of the chain, that is -cis. When they occur on the opposite side, the designation -trans is used, which means across.
d. Teratogenic, from the Greek teras, meaning “monster.” Teratogenic substances cause great malformation in an embryo or fetus.
REFERENCES
1. PA Lehmann and AM Malany AM, Evidence for percutaneous absorption of isotretinoin from the photoisomerization of topical tretinoin. J Invest Dermatol (93), 595–599 (1999)
2. EH Harrisson, WS Blaner, DS Goodman and AC Ross, Subcellular localization of retinoids, retinoid binding proteins, and acyl-CoA retinol acyltransferase in rat liver. J Lipid Res (28), 973–981 (1987)
3. CE Orfanos, CC Zouboulis, B Almond-Roesler and CC Geilen, Current use and future potential role of retinoids in dermatology. Drugs (53),
358–388 (1997)
4. JP Ortonne, Retinoic acid and pigment cells: a review of in-vitro and in-vivo studies. Br J Dermatol. (127 Suppl 41) 43–47 (September 1992)
5. F Ballanger, P Baudry, JM N’Guyen, A Khammari and B Dréno, Heredity:
a prognostic factor for acne. Dermatology 212(2), 145–149 (2006)
6. AR Shalita, JG Smith, LC Parish, et al., Topical nicotinamide compared
with clindamycin gel in the treatment of inflammatory acne vulgaris.
Int J Dermatol (34), 434–437 (1995)
7. A Meyer-Heim, K Landau and E Boltshauser, Treatment of acne with consequences—pseudotumor cerebri due to hypervitaminosis A. Schweiz Rundsch Med Prax. 91(1–2), 23–6 (Jan 9, 2002) (article in German)
8. K Yoshimura et al., Clinical trial of bleaching treatment with 10% all-trans retinol gel. Dermatol Surg. 29(2), 155–156 (Feb 2003)
| Guidelines for Treating Hyperpigmentation A search of the literature reveals a rather standardized treatment in the medical profession for these conditions. Essentially, the products contain hydroquinone from 2–4%, retinoic acid at 0.1% and a high-potency cortisone preparation. Treatment is continued twice daily for 12 or more weeks. There are many preparations for skin lightening and depigmentation, but following are the combinations of ingredients you should seek. A form of vitamin A, such as retinol, retinal, retinyl palmitate, or retinyl acetate, which may vary in concentration from 0.1% to as high as 10%.8 An alpha hydroxy acid, such as lactic, glycolic or malic, or mandelic acid, ascorbic acid (vitamin C) at 2–7%, and a sunscreen with an SPF of at least 15. To this group you can add mulberry extract, arbutin, lemon extract, glucosamine and a host of other ingredients. Remember two things: As you increase retinoid concentration, you will increase irritation; and as you increase the number of ingredients you have, the potential for sensitization and interaction increases. Treatment should be continued for 3–12 months, sometimes longer. Let the client know that these pigmented lesions are produced throughout a long period of time, and will not go away quickly. It is important that the skin care professional has a good understanding of melanogenesis and is able to appreciate the complexity of pigmentation. |
| Guidelines for Treating Aging Skin Start with a 0.3% solution—that is about the middle of the road in strength. Apply at nighttime after cleansing the skin. For 40+ women, I prefer an oil-based cleanser rather than a soap of detergent base. The oil base will not damage the skin’s barrier, but will leave a nice microthin lipid layer that will expedite the entry of the vitamin A preparation. Here again, I prefer the vitamin A ester, retinyl acetate. Following are other preparations available for comparison. The literature provides the following informatione. In the nighttime treatment program, which should be mandatory before 11 pm, add vitamin C topically—it is an antioxidant and collagen booster. Bioflavanoids such as oligomeric proanthocyanidins (OPC) are powerful inhibitors of collagenase, or matrix metalloproteases. These enzymes break down the collagen throughout the body and are responsible for most of the lax skin seen in women. There are many types, but MMP 1 is the worst of the lot. In addition, add carnitine to assist the formation of ATP, the critical energy molecule that supplies the power to run the bodyf. Of course, vitamins D and E should be included. More is being learned about vitamin D as an essential vitamin and hormone for the skin. Make sure a day cream with an SPF of at least 15 is used. It will take many weeks to show improvement, except in the mildest cases. If there is no satisfactory improvement in 4–6 weeks, increase the retinyl acetate to 0.5% and add a facial peel once a month. Do not use trichloroacetic acid; rather, use a milder acid such as salicylic, lactic or malic. Remember that the turnover rate for skin increases with age; because of this, frequent chemical peels are not beneficial at intervals of less than four weeks. You may have to treat your client for life. Aging can be slowed, but not stopped at this stage. You can repair, correct and prevent aging changes, but the process, as of now, is relentless. This is not to say there is no hope for advances that are being made. Even today, women of 30 or 40 years of age can hope to look the same, or even better, in their 70s and 80s. e. The literature refers to the scientific literature, namely periodic journals from which all scientists quote and refer. It is not a specific journal; it is the whole scientific world thousands of journals. f. ATP is adenosine triphosphate, is made in the mitochondria and is essential to most biological reactions. |
| Guidelines for Treating Acne This is a working classification for addressing acne that follows the American Academy of Dermatology recommendations. Mild acne—only comedones. Use a salicylic acid 2–5% cleanser twice daily and follow in the evening with retinol or retinyl acetate, 0.5%, at night. Reduce fatty and sugar-laden foods, and make sure green and yellow vegetables are consumed. Lesions should clear in 3–6 weeks. Follow up with bi-monthly visits for at least six months, then, at six-month intervals. Mild acne with comedones, macules and papules. Follow the same regimen as mild acne with only comedones, but add 2.5–5% benzoyl peroxide. At this stage, infection has started and the Propionibacterium acnes needs to be stopped from making a pustule. Follow weekly with oxygen treatments—a proven topical oxygen or a water-based aerosol of oxygen is ideal. Dry oxygen is not as effective. Getting into the follicle with molecular oxygen is the goal because P. acnes is anaerobic and does not like oxygen. You should see this client every week until the lesions are clear. Papules and pustules. Pustular acne means that the infection has gotten worse. Physicians generally use oral antibiotics at this stage, but you could use all of the aforementioned agents plus 5% niacinamide cream, which is available from some suppliers. Niacinamide at 5% has been shown to be as effective as clindomycin when applied topically.6 If you are not getting a response in a week, the client should be referred to a physician. |










